| Post is a part of a larger series (Uniaxial base): |
If you have ever tried to design a uniaxial base using circle packing method, then you probably know that process is based on the so-called divide and conquer approach. To make the long story short: all crease patterns of uniaxial bases can be divided into the axial polygons. These polygons can then be analysed and solved individually. The only prerequisite is that polygons are convex.
With this in mind, I would strongly advise you, before proceeding further, to read a blog post on the Uniaxial base.
That being said we can proceed further. So, what is the procedure once we manage to define all axial polygons? What is the next step? Well, the next step calls for adding a set of creases into every single axial polygon. These creases, commonly known as origami molecules, will enable axial creases that form a polygon, to coincide when the base is collapsed.
Types of molecules
Origami molecules can be divided into distinct groups based on the shape of axial polygons for which they are intended. Or to be more precise, they are grouped based on the number of sides these polygons have. For instance, for polygons in the shape of a triangle, a triangle molecule is used. This molecule has a quite strange name: a rabbit ear molecule. But despite its strange name, this molecule is a remarkable one. Namely, it can be constructed for any triangle polygon, regardless of its shape and size. Therefore, no other molecule for triangle polygons is needed (If you want to know more about the rabbit ear molecule, feel free to read the blog post on the Rabbit ear).
On the other hand, if we talk about molecules for the quadrangle polygons situation is a bit more complicated. You see, there is more than one type of molecule that will do the job. Some of them are more versatile than the others, but the most frequently used one is so-called Gusset molecule. Its versatility is such that it makes almost all other types of molecules obsolete. But truth be told, such versatility of a Gusset molecule lies in the fact that Gusset molecule is, in fact, a universal molecule in disguise. We can go even further and claim that even the famous rabbit ear is also a universal molecule in disguise. So, the legitimate question arises: what is the universal molecule?

The Universal molecule
By definition, the universal molecule is a set of creases that can be added to the axial polygon of any size and shape, allowing all axial creases of that polygon to coincide when a base is collapsed or folded. Such a generic definition calls for a further explanation, so please stay with me. You will not regret it.
But before we move on, let me point out one important fact.
You see, there is a direct link between the folded base size and the size of its crease pattern. Or to be more precise; the distance between two points on the folded base cannot be larger than the distance between the same points on the crease pattern. That is logical since, for such an odd case to occur, a paper must be stretchable, and I believe that you can agree with me that paper, or at least, the one used for origami is not stretchable. Therefore the previous statement must hold.
With this fundamental question being sorted out, we can proceed further and try to construct a universal molecule from scratch.
Axial polygon definition
Design of every uniaxial base starts with a stick figure. Stick figure represents the model in a sense that it shows all the necessary parts in right proportions.
So, let’s start with a simple example. According to the stick figure in figure 2, we have to pack one river and five circles of various sizes on a square piece of paper. An optimal arrangement of the circles and the river in between them is shown in figure 2.
(If you are not familiar with this concept, fill free to read a blog post on the Relationships between basic elements of an origami model)
The next step is to find positions of all axial creases and consequently to construct the axial polygons.

By definition, axial creases connect circle centres, but not any centre. To connect two circle centres with an axial crease, circles must touch each other. To be exact, a river is allowed to be in between, but any kind of gap is not allowed.
So, let’s try to add axial creases according to that rule. First, let’s examine circles C and D. These circles touch each other therefore we can connect them by adding an axial crease. On the other hand, an axial crease between circles A and D cannot be created, since there is a gap in between. Whenever a gap exists, the path between the circles cannot be called active. Now, I almost hear you saying: what is an active path? Quite simply, if the distance between two points on the origami base is the same as on the crease pattern then we can say that the connection between these two points is an active path. If the distance is larger, meaning there is a gap in between than the path cannot be called active.
So, in figure 2, where axial creases can be created? If we apply the previous rules the answer is obvious. Additional axial creases can be created between circles A-B, B-C, C-D, D-E and finally A-E because paths between them are active.
Under which circumstances is a gap allowed?
Let’s consider two triangles in figure 2. The second one is a well-known rabbit ear, whereas the other is also a rabbit ear but somewhat ill-defined. I have told you in the beginning that for every triangle, only one rabbit ear exists. That implies that only one combination of circles can produce such a triangle. In that combination, all three circles touch each other, making all paths the active one. A favourable result of having all axial creases defined as the active path is a possibility to create an axial polygon that can be collapsed or folded with ease.

What about another triangle?
That triangle is a result of a different combination of circles. But what is very important to notice is that these circles touch each other in only two points instead of three. That implies that the line between points B and C does not represent an active path (a gap exists) therefore, such a triangle can not be considered an axial polygon. But there is an exception.
If you take a good look at figure 3, you will realise that it is possible to collapse even such an ill-defined triangle.
That brings us to another rule. Any polygon can have an inactive path (the side with a gap in it) if the path in question is on the paper edge. For now, remember this as a rule. At the end of this blog post, I will show you this in practice.
Universal molecule construction
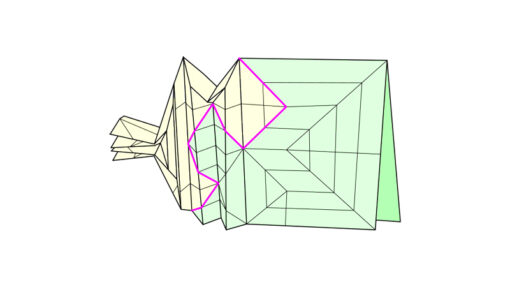
Let’s go back to our example. Here we have an axial polygon the shape of a pentagon which we have to fill in with a bunch of creases in order to make polygon sides to coincide where the base is collapsed.

This pentagon-shaped polygon is indeed an axial polygon because it cannot be divided any further. We could try, but in that case, we will get polygons whose sides are not active because to achieve that, we will have to connect centres between non-adjacent circles.
Procedure for defining creases inside of the polygon is as follows. First, we begin by adding angle bisectors (purple lines), effectively reducing the size of the polygon. The reduced polygon (marked in green) is clearly visible in figure 4. As you can see, it has the same shape as the original polygon. Reduction of the polygon goes until a certain point is reached. At that point the path between two non-adjacent circles’ centres becomes active.

In figure 5, you can see the original axial polygon and reduced one. Now let’s examine similarities as well as the differences between these two polygons. On both polygons, the river is the same, but the circles are a little bit different. They are smaller since they have to fit into the smaller polygon. But what is most important is the fact that circles A and D squeeze the river tightly, effectively making the path between them active. This path is now the active one since there is no gap whatsoever. At this point, obviously, the polygon cannot be reduced any further.

At this point, the reduced polygon can be divided into two smaller polygons along the newly formed active path. If you look at figure 6, one of these newly formed polygons is a triangle for which rabbit ear molecule can be used. Another one is a quadrilateral polygon for which we have to repeat the reduction process once again.

Again, a polygon reduction procedure is performed, until another path becomes active. In this example, the path between circles A and C becomes active, so the reduction procedure has to be stopped once again.

I think you can guess what the next step is. Of course, the polygon again has to be divided into two smaller polygons along the newly defined active path. Both newly formed polygons are triangles for which solution is well known.

Since we resolved both triangles by introducing the rabbit ear molecule, our crease pattern is almost finished. What is left to be done is a crease orientation definition or mountain-valley assignment. The task is fairly easy because in most cases angle bisectors are mountain folds while the lines that divide polygons into two smaller ones are valley folds (figure 10).
The fully collapsed base is available in figure 10 as well.

Polygon reduction process in real life
If you closely followed the reduction procedure, most certainly you have noticed that polygon is always reduced to a certain point. But how do we know when this particular point is reached? According to the definition, the reduction procedure has to be performed until one of the paths becomes active as it was explained in the previous example. The easiest way to do so is to use TreeMaker developed by Robert Lang. This freely available software will do the whole process automatically and accurately.
Unfortunately, there is a problem of folding such a crease pattern. You see, if you try to fold it, you will soon realize that it is almost impossible to transfer these lines onto the paper only by folding. To accurately do so, ReferenceFinder, also developed by Robert Lang, should be used.
Polygon reduction process – additional rules
While performing a polygon reduction procedure, achieving an active path is by no means the only criteria that will end the polygon reduction.
Sometimes, the reduction procedure must be stopped, due to the collision of two, or more bisectors. But, let me show you an example that includes such a situation (figure 11).

In figure 11, you can see an example that includes five circles and a river. Again, the procedure is the same. Reduction of the pentagon size leads to the creation of two simpler polygons. First one, a triangle is not particularly interesting, but the second one, a quadrilateral polygon definitely is (figure 12).

I hope you understand how we come up with the quadrilateral polygon. Now comes the important part.
If we proceed with the reduction procedure of quadrilateral polygon, we will realize that two angle bisectors will converge into the single point before any inactive path becomes active. Nevertheless, we have to stop there since a new triangle polygon is already created (look at figure 13).

The newly formed triangle is like all previous triangles populated with rabbit ear molecule.
That led as to the final step, mountain-valley assignment (figure 14). But before we do that, I would like to make a recap.
Construction of the universal molecule is a very simple and straightforward procedure. Continual reduction of the axial polygon should be performed until one of two events occurred. Either one of the inactive reduced paths become active, or two or more angle bisectors converge into the same point. Result of one of these two events is the creation of simpler polygons. The procedure repeats itself until the whole crease pattern is defined. In other words, until all polygons are reduced into the triangle polygons.
So, let’s go back to our example and see how a fully developed crease pattern, as well as the folded base, look like.

Quite simple, isn’t it?
Putting everything together
Since we know how to construct the universal molecule, I would like to show you the whole theory on a more complex example. An example is artificially constructed, only to point out a few significant problems regarding the universal molecule usage. For a given example, 10 nodes and 4 rivers have to be positioned on the paper. The layout shown in figure 15 is one of the possible solutions. But even though the layout shown represents an almost optimal solution, a large chunk of paper in the middle is inevitably left unused. Such a situation will have undesirable consequences on the final model.
But let’s solve this example first. Then, I believe, you will see why such a large amount of the unused paper in the middle represents a potential problem.

First, we have to find all axial creases that form axial polygons. The task should be fairly easy since we already know that axial creases connect centres of adjacent circles. So let’s find them.

As you can see in figure 16 there are five axial polygons; two triangular polygons, two quadrilateral polygons and one hexagonal polygon. We should start from two identical triangular polygons.
Triangular polygons
What is particularly interesting about them is that one of their sides is not active, there is a gap between circles D-F and A-I. Normally, such a situation is not allowed unless this polygon side is on the paper edge. In our case, there is no doubt that polygon sides are on the paper edges so both polygons can be considered valid axial polygons. Being aware of that, we can without further due implement rabbit ear molecules, as it is shown in figure 17.

Quadrilateral polygons
Quadrilateral polygons are much more interesting because their solution is not known in advance. We have to gradually reduce the polygon until one of the reduced paths becomes active. The reduction procedure is shown in figure 18.

The procedure is carried on until the reduced path between circles B and D becomes active. The quadrilateral polygon is then divided into two triangular polygons for which solution is known; rabbit ear. I don’t think I have to point out that an identical procedure applies to the other quadrilateral polygon too.

Hexagonal polygon
Finally, we come to the most complicated part. To be honest, hexagonal polygons are not that complicated either since only what you have to do is to follow the procedure. After all the procedure is very simple, you have to gradually reduce the axial polygon until one of the reduced paths becomes active. That is all.

When the reduced path between two central circles becomes active, the reduced hexagonal polygon is divided into two quadrilateral polygons. Solution for any quadrilateral polygon is already explained so I would not go into details. After all, our example is the standard one in which reduction of the polygon leads to the creation of two triangular polygons and implementation of rabbit ear molecules.

By solving hexagonal polygon, and by applying the mountain-valley assignment, the final crease pattern is obtained. If you look at the crease pattern in figure 21, you will realize that it is not very complicated, since the number of creases is relatively small. Such a configuration is by all means desirable. Unfortunately, the base obtained by implementing universal molecule is in most cases odd looking. The base is unusually wide. A quick look at the hinge polygons will show you the reason.

If you look at the flap B hinge polygon, you will see that the polygon itself is of very unusual shape. Exactly that odd polygon shape gives the flap B such an unusual width.
The base is deliberately designed in such a way that a large partition of the paper is empty. Since no paper can be left unused some hinge polygons are inflated to occupy that space, leading to the unusually wide flaps. Flap E is a good example.